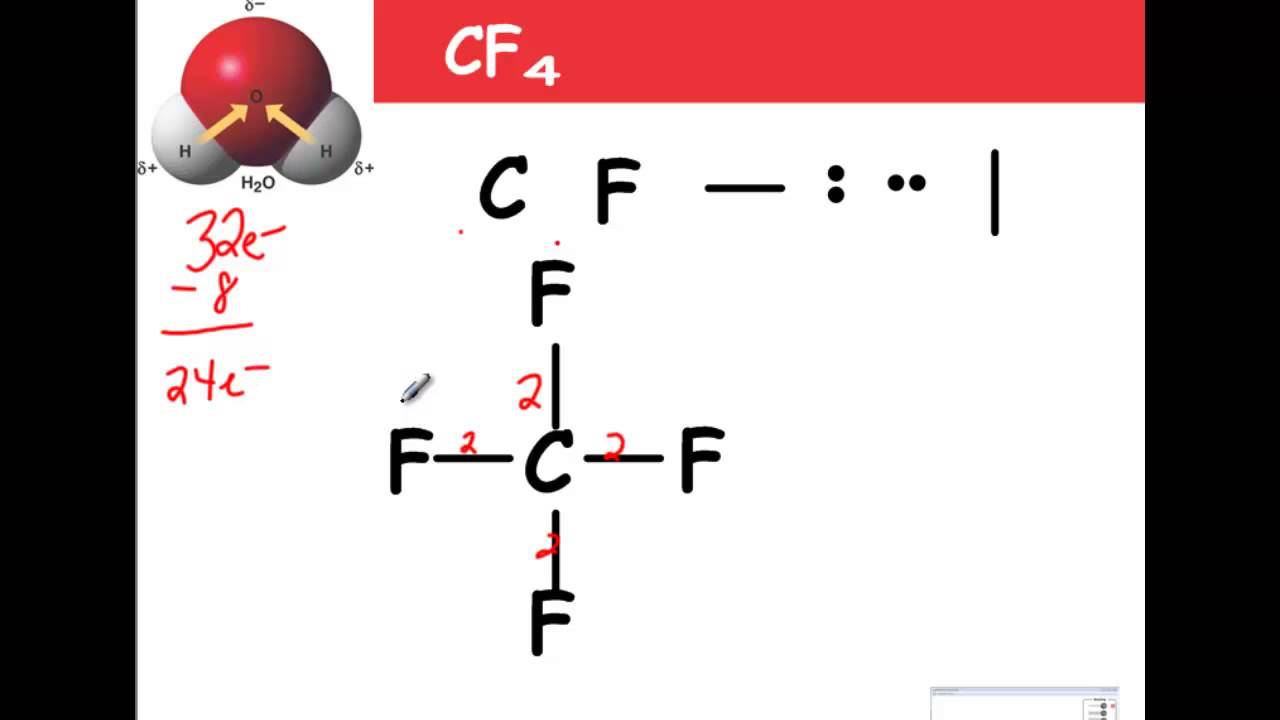
It can be studied in detail with the help of the Valence Shell Electron Pair Repulsion (VSEPR) theory that in the presence of lone pair of electrons, the molecule tends to follow the ideal conditions.Ĭarbon tetrafluoride (CF4) is the AX4E0 type structure where ‘A’ represents carbon, ‘X4’ represents four fluorine atoms and ‘E0’ represents zero lone pair of electrons. 
Now merge all the above-mentioned points and draw the Lewis structure of the carbon tetrafluoride (CF4) molecule:Ĭarbon Tetrafluoride (CF4) Molecular GeometryĬF4 is a Penta-atomic molecule having a bond angle of 109.5° which gives rise to a tetrahedral shape. Only a single covalent shared bond is formed between the participating atoms. Step 4: Look for the type of bonds formed in each carbon tetrafluoride (CF4) molecule (single, double, or triple bond). They tend to make the maximum number of bonds with other participating atoms. 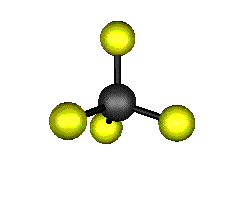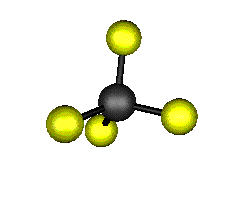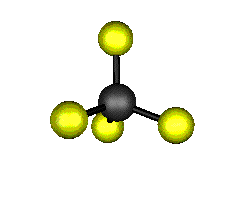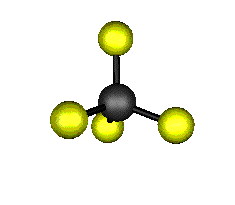
Step 3: Look for the central atom in each carbon tetrafluoride (CF4) molecule: Usually, the central atom is the one that is present as the single entity within the molecule. Step 2: Find how many more valence electrons are needed by each carbon tetrafluoride (CF4) molecule: It is 8 as 4 more are needed by the carbon atom and one each by a fluorine atom. Step 1: Find the total number of valence electrons each carbon tetrafluoride (CF4) molecule has: It is 32 as 4 are coming from the carbon atom and 7 are coming from each fluorine atom. 
Now, let’s learn to draw the Lewis structure of carbon tetrafluoride (CF4) with steps: Even the slightest excitation by any nearby atom, these electrons readily bonds with them to form a new compound having new properties altogether. Only these electrons participate in the bond formation as they are present farthest from the nucleus.ĭue to this, the force of attraction by the nucleus does not reach effectively to these electrons. Here it is important to know first what the valence electrons are? The electrons available in the outermost shell of an atom are called valence electrons. This makes the valence electrons in the fluorine atom 7. The valence electrons are also those which are present in the highest principal energy levels which are 2s and 2p for fluorine. Whereas, in the case of fluorine, its atomic number is 9 for which the electronic configuration is 1s2 2s2 2p5.īesides this, we know chemistry is full of exceptions, so is the case with the fluorine atom. This makes the valence electrons in carbon 4. The atomic number of the carbon is 6 which makes its electronic configuration 1s2 2s2 2p2.Īs we know, the p shell needs to accommodate 6 valence electrons in its outermost shell, there is a dearth of 4 valence electrons. It is the Lewis structure only by which we can study the molecular orbital diagram of a molecule.Īs we know, to study the Lewis structure of any compound, we first need to analyze the structure of participating atoms. It is important to know in which shell the electrons are orbiting along with their energy levels to further study the hybridization of a molecule. Moreover, the diagram helps with determining how these valence electrons participate in the bond formation either by getting accepted or donated to another atom. The Lewis structure is a diagrammatic representation of determining the position of valence electrons within an atom. Where this compound is used in electronic microfabrication, it is also one major greenhouse gas as it has a lifetime of more than 50,000 years making it a sound contributor to global warming.Ĭonclusion Carbon Tetrafluoride (CF4) Lewis Structure This compound being slightly soluble in the water is used as an important refrigerant at a low temperature. When dichlorodifluoromethane reacts with hydrogen fluoride, it produces carbon tetrafluoride and hydrogen chloride.īesides this, being a highly stable compound, the thermal decomposition of carbon tetrafluoride produces toxic gases of carbonyl fluoride and carbon monoxide. There exist many ways of preparing carbon tetrafluoride but the most commons industrial method is using hydrogen fluoride. It is available in a colorless and inflammable gaseous state and usually transported in liquid form under very high pressure. Moreover, the compound is also called tetrafluoromethane as it belongs to the group of fluoromethanes.Ĭarbon tetrafluorides are widely used to study fluorine chemistry to prepare organofluorine compounds. It is a well-known haloalkane or halomethane having a high bond strength between the carbon and fluorine atoms becoming quite a stable compound. CF4 is the molecular formula of Carbon Tetrafluoride and is the simplest fluorocarbon of all.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
